Next-Generation Invasive Lung Ventilation
The Global Ventilation Crisis
Standard mechanical ventilation is a lifesaving but "necessary evil." By using positive pressure to force air into the lungs, current ventilators bypass natural respiratory mechanics, leading to Ventilator-Induced Diaphragmatic Dysfunction (VIDD) in over 60% of patients within 48 hours. This results in:
The "Weaning" Bottleneck: Patients become physically unable to breathe independently, leading to prolonged ICU stays.
Economic Burden: ICU bed costs exceed £3,000/day; delayed weaning costs global healthcare systems billions annually.
Patient Morbidity: High sedation requirements prevent swallowing, speech, and early rehabilitation.
Shifting the Paradigm
As per our finding with industrial discussions, the challenge was to transition from External Positive Pressure to Internal Negative Pressure augmentation. Hence, we engineered a biocompatible, implantable "muscle" that can take over or assist the diaphragm’s natural contraction, allowing the lungs to expand as nature intended—even when the biological muscle is paralyzed or weak.
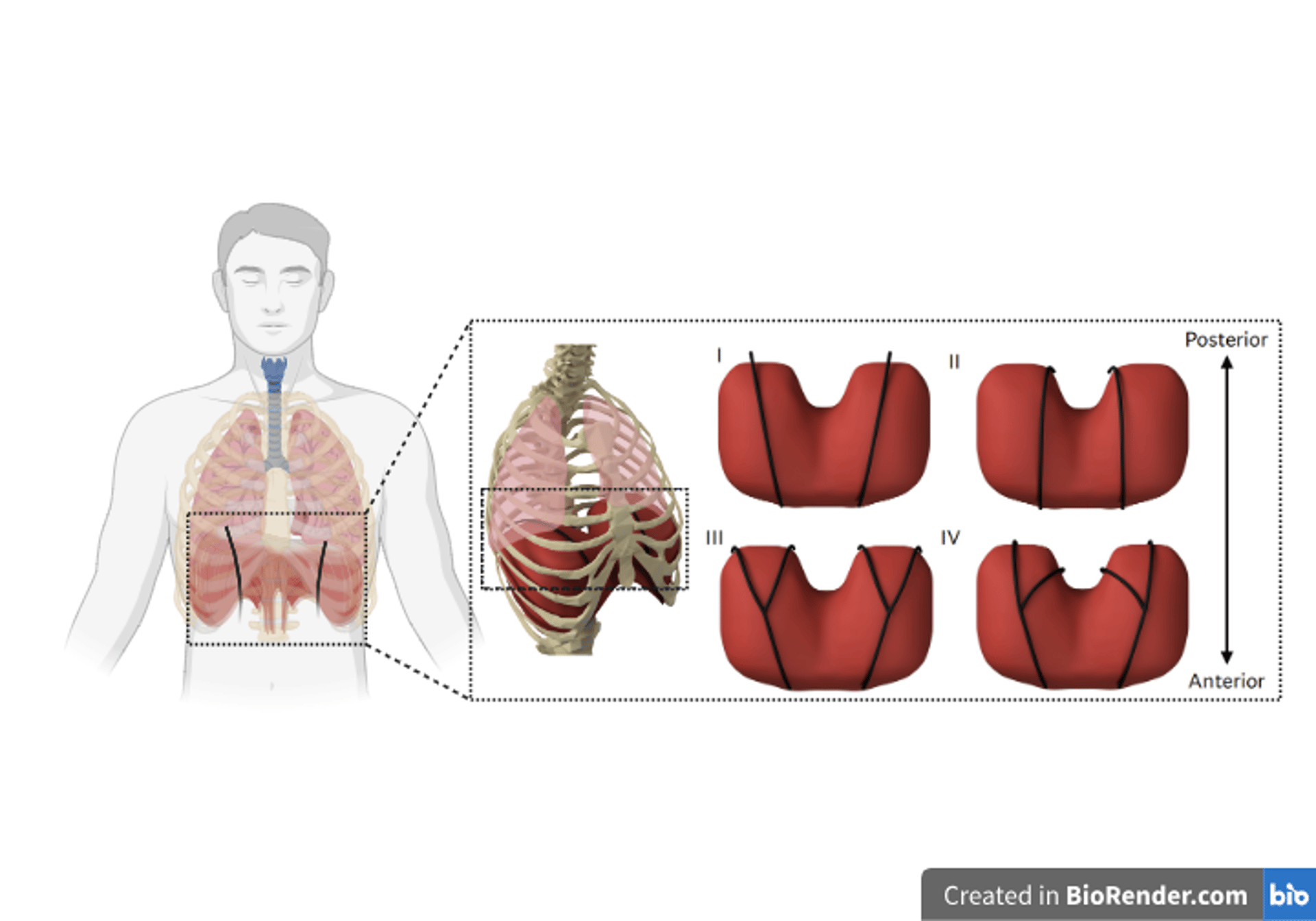
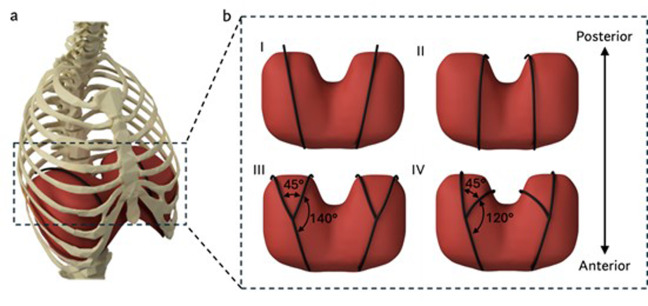
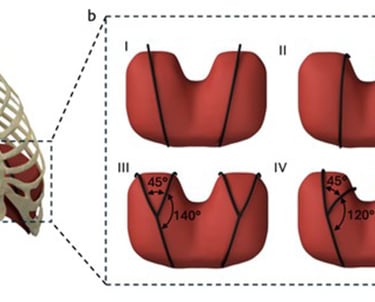
(a) A rendered representation of the actuators (in black) superior to the diaphragm for augmenting ventilation. (b) Rendered representations of the two actuator geometries (black) and their configurations. (I) Straight lateral. (II) Straight medial. (III) Branched wide. (IV) Branched narrow.
Engineering a Bio-Mimetic Solution
We led the development of a proprietary Soft Robotic Implantable Actuator (SRIA):
High-Fidelity Modeling: Developed computational "Digital Twins" of the human thorax to optimize robotic-tissue interaction.
Advanced Biofabrication: Utilized multi-material 3D bioprinting to create actuators with non-linear elasticity, matching the mechanical impedance of human tissue.
Indigenous Testing Infrastructure: Established a world-class thoracic simulation test-bed to validate device performance under realistic physiological loads.
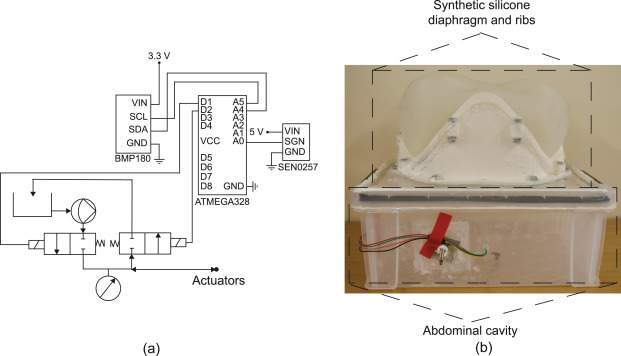
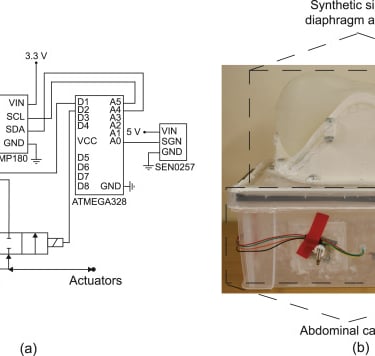
The experimental setup showing the electrohydraulic schematics (a) used to control and measure the pressures of the actuators and abdominal cavity, and (b) the test bed showing the synthetic silicone diaphragm clamped to the ribs, which in turn are attached to the abdominal cavity.
Peer-Reviewed Validation
A Scalable Medical Device Pipeline
Technical Proof-of-Concept: Demonstrated the world’s first soft-robotic implant capable of restoring tidal volumes without positive air pressure.
De-Risking Translation: Built an end-to-end pipeline from digital design to pre-clinical porcine models, significantly reducing the "Valley of Death" for new MedTech.
Industry Readiness: Secured partnerships with leading bioprinting firms (Regemat 3D) to ensure the scalability of the manufacturing process.
Impact: This research provides a roadmap for a new class of "Active Implants" that could reduce ICU weaning times by 30-50%, drastically improving patient outcomes and hospital throughput.
Our Team


Manish Chauhan
Principal investigator


Christopher Michael Hofmair
Meng researcher


Dr. Kunal Bhakhri
Consultant Thoracic Surgeon
MEd, FRCS (CTh)
Honorary Visiting Professor at University of York - Clinical collaborator


Dr. Laura Fitton
Programme Director for MSc Human Anatomy and Evolution, Lecturer in Anatomical and Human Sciences
Contact
Reach out for collaborations or questions.
Phone
manish.chauhan[at]york.ac.uk
+44 (0) 1904 32 2361
© 2025. All rights reserved.
